Chemical Reactions and Balancing in Alignment with the NGSS
|
HS-PS1.B-"Chemical processes, their rates, and whether or not energy is stored or released can be understood in terms of the collisions of molecules and the rearrangements of atoms into new molecules, with consequent changes in the sum of all bond energies in the set of molecules that are matched by changes in kinetic energy."
HS-PS1-7-"Use mathematical representations to support the claim that atoms, and therefore mass, are conserved during a chemical reaction."
|
In this lesson, students will use an exploratory PhET activity to learn how a chemical reaction occurs. Students will also be introduced to the Law of Conservation of Mass/Matter, and how this applies to balancing Chemical Equations. The students will practice balancing Chemical Equations with a worksheet and homework.
|
Introduction to Chemical Reactions and Balancing Lesson Plan
Start of Class:
To start the class, the goals for the day will be listed on the board, and gone over by the teacher. The student goals for this lesson are:
1) Students can state how a Chemical Reaction occurs
2) Students can state the Law of Conservation of Mass/Matter
3) Students can us the Law of Conservation of Mass/Matter to balance Chemical Equations
The teacher will also make class announcements such as the class activities for the day and describe to the students what the performance expectations are for the day. The class activities for the day will include a Youtube video, work in the Computer Lab, a small amount of note taking, finishing up with group work time. The teacher should tell the students that they are expected to be attentive, and on task throughout the lesson.
Introduction of Lesson:
This lesson will be introduced via a Youtube Video. To access this video, please click the link below. Prior to showing the video, the teacher should make the students aware of what they are to be looking for in the video. These things include: particle collisions, kinetic energy, activation energy, potential energy, and Chemical Reactions.
Chemical Reactions VideoVideo from Classroom Video Ltd, www.classroomvideo.co.uk
Following the video, the teacher should use an informal assessment to see what the class got out of the video, and to reiterate the key concepts.
Lesson Instruction:
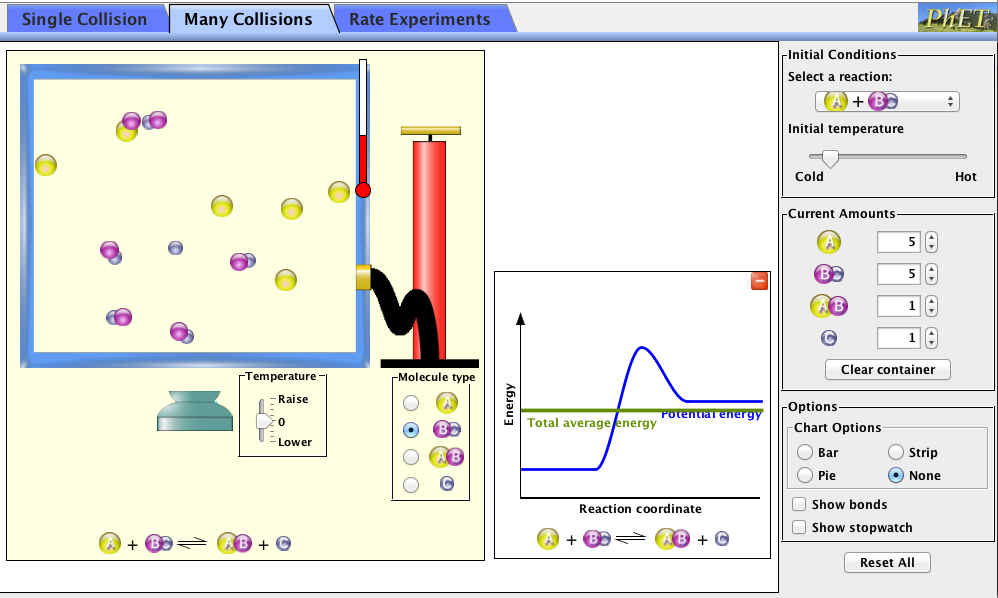
After the conversation about the video, the teacher will tell the students that they will be exploring the ideas from the video in an online simulation. The teacher will show students how to access the simulation (linked below), and a little bit how to use it. The teacher will draw the students' attention to the reaction coordinate graph as seen in the simulation, and tell them that they will be looking more closely at those tomorrow, but the students should notice what happens to the graph as they play with the simulation.
"Reactions and Rates" Simulation
To start the class, the goals for the day will be listed on the board, and gone over by the teacher. The student goals for this lesson are:
1) Students can state how a Chemical Reaction occurs
2) Students can state the Law of Conservation of Mass/Matter
3) Students can us the Law of Conservation of Mass/Matter to balance Chemical Equations
The teacher will also make class announcements such as the class activities for the day and describe to the students what the performance expectations are for the day. The class activities for the day will include a Youtube video, work in the Computer Lab, a small amount of note taking, finishing up with group work time. The teacher should tell the students that they are expected to be attentive, and on task throughout the lesson.
Introduction of Lesson:
This lesson will be introduced via a Youtube Video. To access this video, please click the link below. Prior to showing the video, the teacher should make the students aware of what they are to be looking for in the video. These things include: particle collisions, kinetic energy, activation energy, potential energy, and Chemical Reactions.
Chemical Reactions VideoVideo from Classroom Video Ltd, www.classroomvideo.co.uk
Following the video, the teacher should use an informal assessment to see what the class got out of the video, and to reiterate the key concepts.
- What is a Chemical Reaction
-Answers should discuss something along the lines of atoms rearranging to form new molecules - How do Chemical Reactions occur?
- Answers should involve molecule or particles colliding with one another, as well as the particles having enough energy for a reaction to take place. - How can the rate of a Chemical Reaction be changed? (if students are struggling with this, refer back to the video when activation energy was talked about
- Answers should involve increasing the energy of particles so that they reach the activation energy by increasing temperature or concentration. Answers can also include stirring liquids or increasing the pressure of gases and talk of kinetic and potential energy of particles.
Lesson Instruction:
After the conversation about the video, the teacher will tell the students that they will be exploring the ideas from the video in an online simulation. The teacher will show students how to access the simulation (linked below), and a little bit how to use it. The teacher will draw the students' attention to the reaction coordinate graph as seen in the simulation, and tell them that they will be looking more closely at those tomorrow, but the students should notice what happens to the graph as they play with the simulation.
"Reactions and Rates" Simulation
Prior to moving to the computer lab, the students will be given a worksheet to complete as they use the PhET simulation. The students will be given about 10 minutes to play with the simulation and answer the questions on the worksheet. This sheet will then be collected and used as a form of assessment to see how the students are viewing the material on how chemical reactions occur.
Once the students have arrived back in the classroom, the teacher will ask the students to settle back into their desks, and take out a notebook. The teacher will go over a few PowerPoint slides on how to write a Chemical Equation, and how to balance them. The PowerPoint includes some examples of how to balance a Chemical Equations. The teacher should go over these with the class, giving the class time to attempt the problems prior to giving the answers.
Once the PowerPoint is finished, the teacher will hand out a Balancing Chemical Equations worksheet. The students will have the remainder of the class period to work on this worksheet (in groups if they want to). The students will be told that if they do not finish the worksheet, it is to be done tonight for homework and that the more difficult questions will be gone over tomorrow in class. This worksheet will also be collected as a form of assessment.
Assessments/Checks for Understanding:
The assessments for this lesson comes from multiple areas. The first being the conversation the teacher has with the students after watching the Youtube video. This conversation is to check for the understanding that a chemical reaction occurs when two particles collide with the correct amount of energy (overcoming the activation energy). This concept is also assessed with the PhET activity.
The students concept of the Law of Conservation of Mass/Matter is assessed with the the PowerPoint notes, and the Balancing Chemical Equations worksheet. The teacher should be looking to see that students can state that matter is neither created nor destroyed during a chemical reaction.
Finally, the teacher will assess whether or not students can use the Law of Conservation of Mass/Matter to balance Chemical Equations with the Balancing Chemical Equations worksheet that is assigned for homework and the example problems in the PowerPoint. The teacher should see that students can coefficients and subscripts in Chemical Equations in order to balance the Chemical Equation.
Closure/Wrap-Up/Review:
The teacher will call the class back together to wrap up the lesson in the last few minutes of the period. The teacher will review the goals for the day that were written on the board at the beginning of the lesson. Relate these goals to the lesson, and where in the lesson they were covered. Give a brief introduction to what will be covered into tomorrows class -- wrap up balancing, talk about energy of Chemical Reactions and how it is released or consumed (exothermic and endothermic). Make sure to relate these topics to what was covered today with the energy graphs in the PhET simulation, and the discussion of energies (activation, kinetic, and potential) in the video. At this point, the teacher will make sure all of the students have handed in the appropriate materials (PhET worksheet, and Balancing Chemical Equations worksheet, if finished). Make sure the students know that the Balancing worksheet is to be finished as homework, if they did not complete it in class.